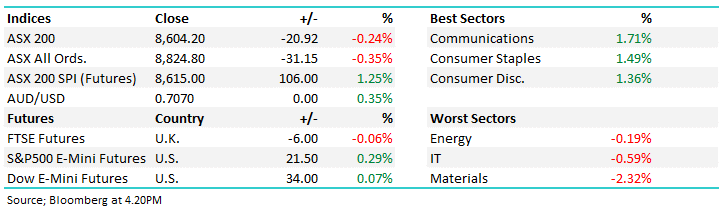
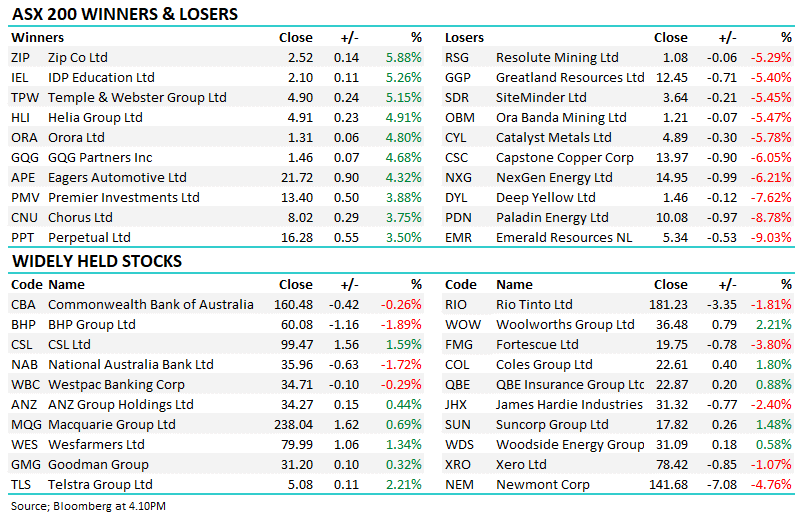
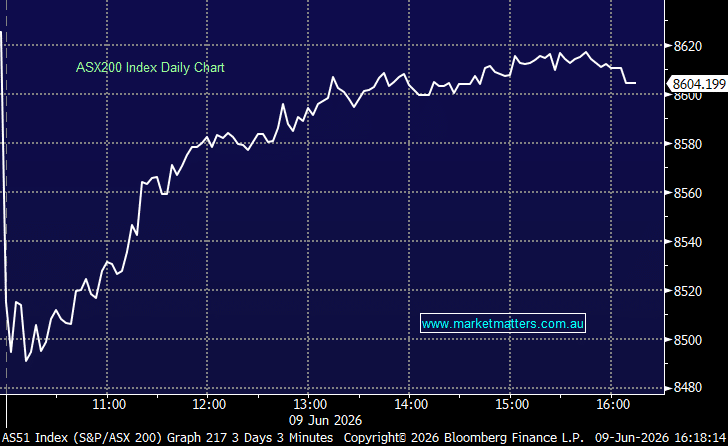
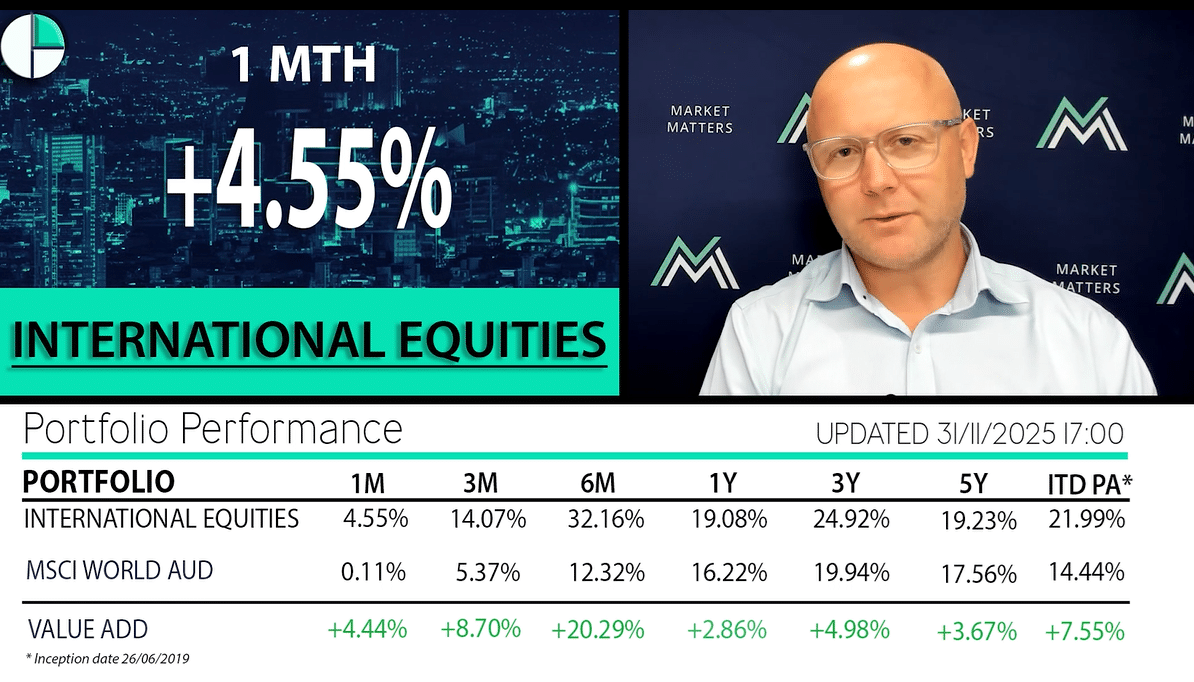
14 DAYS FREE ACCESS
Join Market Matters and get full access
to all our Opinion + Action
TLX $13.39
20 MINUTE DELAYED
i
PERFORMANCE
i
| SHARE PRICE ACTIVITY | |
|---|---|
| Last (Price) | 13.39 |
| Change | 0.08 (0.60) |
| Bid / Ask | 13.37 - 13.48 |
| Volume | 2,140,549 |
| Turnover | 28,473,408 |
| Open | 12.95 |
| Day Range | 12.80 - 13.50 |
| VWAP | 13.29 |
| Prev Close | 13.31 |
| Last Trade | 09/06 4:10pm (AEST) |
i
| KEY INFORMATION | |
|---|---|
| Sector | Healthcare |
| Shares Issued | 280,812,404 |
| NTA/Share | 0.00 |
| Last Dividend | 0.00 |
| Dividend Ex Date | 0 |
| Dividend Pay Date | 0 |
i
| CURRENT () | |
|---|---|
| Revenue | |
| EBITDA | |
| Profit | |
| Earnings Per Share (EPS) | 0 |
| Dividend Per Share (DPS) | 0 |
| Yield % | 0.00 |
| Market Cap | 0 |
| Price to Earnings Ratio (P/E) | 0 |
i
TARGET PRICE FORECAST
i
| ESTIMATES () | |
|---|---|
| Revenue | 0.00 |
| EBITDA | 0.00 |
| Profit | 0.00 |
| Earnings Per Share (EPS) | 0.00 |
| Dividend Per Share (DPS) | 0.00 |
i
BROKER CONSENSUS RECOMMENDATION
BUY
SELL
Target Price
Lt Growth Rate
Recommendation ()
ACTION

Business Summary
Telix Pharmaceuticals Limited is a radiopharmaceutical company focused on the development of diagnostic and therapeutic products using Molecularly Targeted Radiation (MTR) to treat cancer. In MTR therapy, a radionuclide is attached to a targeting agent such as a small molecule or antibody. This targeting agent specifically binds to tumors and delivers a radioactive payload in a selective way. The radioactive payload can either be diagnostic (for imaging) or therapeutic, or both. Its lead programs include TLX591-CDx (Illuccix) / TLX591 for diagnosis and treatment of metastatic castrate-resistant prostate cancer, TLX250-CDx/TLX250 for diagnosis and treatment of renal (kidney) cancer, TLX101-CDx / TLX101 for diagnosis and treatment of glioblastoma (brain cancer), and TLX66-CDx (Scintimun) / TLX66 for the treatment of bone marrow conditioning and rare diseases.
Relevant suggested news and content from the site